Cryogenics is a field of science and engineering that delves into the behavior and utilization of materials at extremely low temperatures. At such extreme cold temperatures, substances exhibit unique properties, making them suitable for specific applications. Cryogenic materials and phenomena occur at temperatures below -150°C. Cryogenic environments are often referred to in Kelvins (K) or even milliKelvins (mK).
Cryogenic liquids
One of the most crucial aspects of cryogenics lies in the study of cryogenic liquids. These are substances that remain in a liquid state at very low temperatures and are commonly used in various industries including medical, pharmaceutical, chemical, semiconductor, food and beverage, and more. The proper handling and storage of cryogenic liquids are of paramount importance due to their extreme cold and potential safety hazards.
Material behavior at cryogenic temperatures
Aside from undergoing phase change to convert from a gas to a liquid, materials experience significant changes in their physical properties at cryogenic temperatures. Some key alterations include increased thermal conductivity, reduced electrical resistivity, and altered mechanical properties.
- Liquid nitrogen is one of the most common cryogenic fluids, playing a massive role across medical, pharmaceutical, and biotechnology industries that require cryofreezers to store temperature-sensitive samples and other materials. It is even used in surgical applications including thermal ablation therapies.
- Liquid hydrogen is undergoing significant research and development, particularly in electric vehicle applications including long haul trucking. Liquid hydrogen is also used in the aerospace sector in the operation of rockets, satellites, and space exploration vehicles. Liquid hydrogen and other cryogenic fluids are used to propel and cool these systems.
- Liquid helium is the coldest of all cryogenic liquids, with a boiling point of -270 degrees Celsius (3 Kelvin). It is used in specialized applications including dilution refrigerators for quantum computing.
Why thermal insulation is essential for cryogenic systems
Thermal insulation is necessary to store cryogenics in liquid form while maintaining safe temperatures and pressures. To maintain ultra-cold temperatures, cryogenic systems use insulation to minimize incoming heat. Without insulation, thermal energy enters the tanks and piping systems via convection, conduction, and radiation.
If the temperature increases past the boiling point of the stored fluid, the liquid will begin to vaporize. As vapor collects, pressure increases inside the structure. Pressure release valves are important components that prevent internal pressure from reaching dangerous levels.
Vacuum Insulation
Vacuum insulation is considered the industry-standard thermal barrier for cryogenic storage, piping, and other systems. Vacuum insulation is often combined with superinsulation, also referred to as multi-layer insulation (MLI).
Vacuum Jacketed Hoses
Vacuum jacketed hoses are critical components in cryogenic systems. They use a vacuum-sealed jacket to thermally insulate the fluids and maintain their extremely low temperatures.
Multi-layer insulation (MLI)
MLI consists of a series of thin, reflective surfaces and helps to reduce radiation heat transfer. Some common multi-layer insulations are made of materials such as aluminized Mylar, separated by layers of nylon or silk netting. Other multi-layer insulations use proprietary materials and technologies.
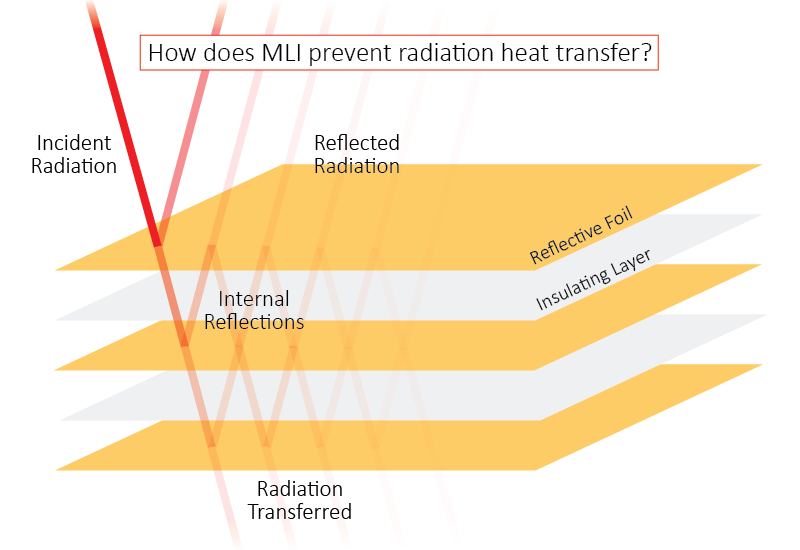
The insulation performance of the MLI depends in part on the vacuum level of the surrounding environment. At a high vacuum level, there will be minimal air between the layers. Air contributes to conductive and convective heat transfer, so less air means less heat passing through the material. That’s why MLI is typically placed within the vacuum space of a double-walled, vacuum insulated container.
Read more:

Insulon® smooth-bore options for thermal process fluid transfer



